Adsorptive removal of Nickel(II) and Chromium(III) from aqueous solutions onto egg shell powder
DOI:
https://doi.org/10.14295/bjs.v5i3.815Keywords:
metal oxide, adsorption process, environmental remediation, aqueous solution, low-cost biosorbentAbstract
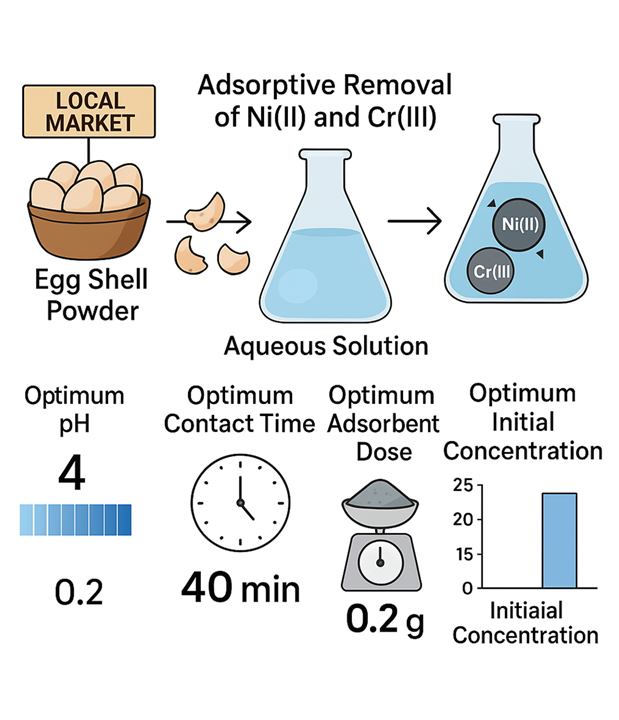
In the current research work, eggshell powder was utilized as an adsorbent for the adsorptive removal of Ni (II) and Cr (III) in a batch process. Eggshells were purchased from a local market in Peshawar, Pakistan. For the adsorption of the adsorbate Ni (II) and Cr (III) from the aqueous solution, the biosorbent, i.e., Egg Shell Powder, was utilized. Different parameters, such as pH, contact time, initial concentration, and adsorbent dose, were optimized. The value of qe and % removal was determined using the optimal conditions. The optimum pH for the adsorption of both Ni (II) and Cr (III) was found to be 4 and 9, respectively. The optimum contact times for Ni (II) and Cr (II) were 40 and 30 minutes. The optimum adsorbent dose for Ni (II) and Cr (III) was 0.2 & 0.1g. The (qe) was determined for Ni (II) and Cr (III) at 25 °C with the increase in concentration of adsorbate. The optimum initial concentration for Ni (II) and Cr (III) was found to be 20 ppm and 40 ppm, respectively.
References
Abatan, O. G., Alaba, P. A., Oni, B. A., Akpojevwe, K., Efeovbokhan, V., & Abnisa, F. (2020). Performance of eggshells powder as an adsorbent for adsorption of hexavalent chromium and cadmium from wastewater. Discover Applied Sciences, 2(12), 1996. https://doi.org/10.1007/s42452-020-03866-w DOI: https://doi.org/10.1007/s42452-020-03866-w
Ahmed, M. M., & Abu-Elsaad, N. (2024). Exploring the magnetic behavior of ferrites: from diamagnetism to superparamagnetism. Condensed Matter. https://doi.org/10.48550/arXiv.2406.10599
Al-Ghouti, M. A., Li, J., Salamh, Y., Al-Laqtah, N., Walker, G., & Ahmad, M. N. M. (2010). Adsorption mechanisms of removing heavy metals and dyes from aqueous solution using date pits solid adsorbent. Journal of Hazardous Materials, 176(1-3), 510-520. https://doi.org/10.1016/j.jhazmat.2009.11.059 DOI: https://doi.org/10.1016/j.jhazmat.2009.11.059
Ali, H., Khan, E. J. T., & Chemistry, E. (2018). What are heavy metals? Long-standing controversy over the scientific use of the term ‘heavy metals’–proposal of a comprehensive definition. Toxicological & Environmental Chemistry, 100(1), 6-19. https://doi.org/10.1080/02772248.2017.1413652 DOI: https://doi.org/10.1080/02772248.2017.1413652
Aljamali, N. M., Khdur, R., & Alfatlawi, I. (2021). Physical and chemical adsorption and its applications. International Journal of Thermodynamics and Chemical Kinetics, 7(2), 1-8. https://doi.org/10.37628/IJTCK
Amir, J., Muhammad, S., Kashif, M., Khan, A. A., Gul, M., Sun, H., Shah, M., Azizi, S., & Maaza, M. (2024). Synthesis, characterization and dielectric properties evaluation of NiO-Co3O4 nanocomposite. Journal of the Iranian Chemical Society, 22, 1-10. https://doi.org/10.1007/s13738-024-03129-0 DOI: https://doi.org/10.1007/s13738-024-03129-0
Angon, P. B., Islam, M. S., Das, A., Anjum, N., Poudel, A., & Suchi, S. A. J. H. (2024). Sources, effects and present perspectives of heavy metals contamination: Soil, plants and human food chain. Heliyon, 10(7), e28357. https://doi.org/10.1016/j.heliyon.2024.e28357 DOI: https://doi.org/10.1016/j.heliyon.2024.e28357
Balali-Mood, M., Naseri, K., Tahergorabi, Z., Khazdair, M. R., & Sadeghi, M. (2021). Toxic mechanisms of five heavy metals: mercury, lead, chromium, cadmium, and arsenic. Frontiers in Pharmacology, 12, 643972. https://doi.org/10.3389/fphar.2021.643972 DOI: https://doi.org/10.3389/fphar.2021.643972
Barceloux, D. G. (1999). Nickel. Journal Toxicology and Clinical Toxicology, 37(2), 239-258. https://doi.org/10.1081/clt-100102423 DOI: https://doi.org/10.1081/CLT-100102423
Bello, O. S., Agboola, O. S., & Adegoke, K. A. (2023). Sources of various heavy metal ions. Heavy metals in the environment: Management strategies for global pollution, Chapter 2, 59-69. https://doi.org/10.1021/bk-2023-1456.ch004 DOI: https://doi.org/10.1021/bk-2023-1456.ch004
Dawra, N., & Dabas, N. (2024). Advances in spectrophotometric determination of Chromium (III) and Chromium (VI) in water: a review. International Journal of Environmental Analytical Chemistry, 104(13), 2994-3015. https://doi.org/10.1080/03067319.2022.2076224 DOI: https://doi.org/10.1080/03067319.2022.2076224
Duffus, J. H. (2001). “Heavy metals”– A meaningless term. De Gruyter Brill, 74(5), 793-807. DOI: https://doi.org/10.1351/pac200274050793
Fiset, J.-F., Blais, J.-F., & Riveros, P. A. (2008). Review on the removal of metal ions from effluents using seaweeds, alginate derivatives and other sorbents. Journal of Water Science, 21(3), 283-308. https://doi.org/10.7202/018776ar DOI: https://doi.org/10.7202/018776ar
Genchi, G., Carocci, A., Lauria, G., Sinicropi, M. S., & Catalano, A. (2020). Nickel: Human health and environmental toxicology. International Journal of Environmental Research and Public Health, 17(3), 679. DOI: https://doi.org/10.3390/ijerph17030679
Gul, M., Kashif, M., Muhammad, S., Azizi, S., & Sun, H. (2025). Various methods of synthesis and applications of gold-based nanomaterials: A detailed review. Cristal Growth & Design, 25(7), 2227-2266. https://doi.org/10.1021/acs.cgd.4c01687 DOI: https://doi.org/10.1021/acs.cgd.4c01687
Harasim, P. (2018). Nickel resources and sources. Nickel in soils and plants. In: Heavy metals, their environmental impacts and mitigation, Chapter, 87-104. DOI: https://doi.org/10.1201/9781315154664-4
Hussain, A., Madan, S., & Madan, R. (2021). Removal of heavy metals from wastewater by adsorption. Chapter 10, 1-24. DOI: https://doi.org/10.5772/intechopen.95841
Hwang, H.-M., Fiala, M. J., Park, D., & Wade, T. L. (2016). Review of pollutants in urban road dust and stormwater runoff: part 1. Heavy metals released from vehicles. International Journal of Urban Sciences, 20(3), 334-360. https://doi.org/10.1080/12265934.2016.1193041 DOI: https://doi.org/10.1080/12265934.2016.1193041
Igwe, J., & Abia, A. A. (2006). A bioseparation process for removing heavy metals from waste water using biosorbents. African Journal of Biotechnology, 5(12), 1167-1179.
Kalsoom, U., Khan, S., Kashif, M., Yaseen, H. S., Hussain, S. A., Azizi, S., & Maaza, M. (2025). MXene-based hybrid composites for lithium-ion batteries: advances in synthesis strategies and electrochemical performance. Ionics, 31(10), 10053-10073. https://doi.org/10.1007/s11581-025-06628-z DOI: https://doi.org/10.1007/s11581-025-06628-z
Kashif, M., Jawad, M., Khan, A. A., Sun, H., Ullah, K., & Fakayode, O. (2024). Fe/Ti-codoped strontium oxide nanoparticles for enhanced photocatalytic degradation of methyl orange. Journal of Applied Research in Water and Wastewater, 11(1), 8-14.
Kashif, M., Khan, A. A., Sun, H., Kamal, J., Shah, M. I. A., Hussain, S., & Ahmad, T. (2024). Synthesis and characterization of Fe-doped CuO nanoparticles: Catalytic efficiency in crystal violet dye degradation and exploration of electrical properties. Brazilian Journal of Science, 3(8), 1-18. https://doi.org/10.14295/bjs.v3i8.601 DOI: https://doi.org/10.14295/bjs.v3i8.601
Kashif, M., Muhammad, S., Ali, A., Ali, K., Khan, S., Zahoor, S., & Hamza, M. (2023). Bismuth oxide nanoparticle fabrication and characterization for photocatalytic bromophenol blue degradation. Journal of Xi'an Shiyou University, 19(07), 521-544.
Khan, M., Sun, X., Kashif, M., Zada, A., Azizi, S., Ragab, A. H., Taher, M. A., Abbas, Q., & Shehzad, K. (2025). Exploration of bismuth-based nanomaterials: From fundamental concepts to innovative synthesis techniques and emerging applications. Coordination Chemistry Reviews, 538, 216687. https://doi.org/10.1016/j.ccr.2025.216687 DOI: https://doi.org/10.1016/j.ccr.2025.216687
Khan, S., Kalsoom, U., Kashif, M., Hussain, S. A., Gul, M., Azizi, S., & Maaza, M. (2025). Smart and sustainable microplastic removal: Hybrid systems, bio-inspired technologies, real-time sensing, and policy integration. Water, Air, & Soil Pollution, 236(14), 900. https://doi.org/10.1007/s11270-025-08468-z DOI: https://doi.org/10.1007/s11270-025-08468-z
Lewis, D. (1988). A study of the factors affecting biosorption column operation: The University of Manchester (United Kingdom).
Liu, J. P., Willard, M., Tang, W., Brück, E., de Boer, F., Liu, E., Liu, J., Felser, C., Fecher, G., Wollmann, L., Isnard, O., Burzo, E., Liu, S., Herbst, J. F., Hu, F., Liu, Y., Sun, J., Shen, B., & de Visser, A. (2021). Metallic magnetic materials. Handbook of Magnetism and Magnetic Materials, 693-808. https://doi.org/10.1007/978-3-030-63210-6_16 DOI: https://doi.org/10.1007/978-3-030-63210-6_16
Madhav, S., Mishra, R., Kumari, A., Srivastav, A., Ahamad, A., Singh, P., Ahmed, S., Mishra, P. K., & Sillanpää, M. (2024). A review on sources identification of heavy metals in soil and remediation measures by phytoremediation-induced methods. International Journal of Environmental Science and Technology, 21(1), 1099-1120. https://doi.org/10.1007/s13762-023-04950-5 DOI: https://doi.org/10.1007/s13762-023-04950-5
Milojković, J. V., Stojanović, M. D., Mihajlović, M. L., Lopičić, Z. R., Petrović, M. S., Šoštarić, T. D., & Ristić, M. Đ. (2014). Compost of aquatic weed Myriophyllum spicatum as low-cost biosorbent for selected heavy metal ions. Water, Air, & Soil Pollution, 225(4), 1927. https://doi.org/10.1007/s11270-014-1927-8 DOI: https://doi.org/10.1007/s11270-014-1927-8
Романова, Л. Г. (2017). Metals. Оренбург, Издательство ОГПУ.
Prasetyo, I. (1992). Removal of toxic metals from aqueous solutions by biosorption. Thesis, Department of Chemical Engineerin, McGill University, Montreal, 1-117.
Rekha, D., Kumar, J. D., Jayaraj, B., Lingappa, Y., & Chiranjeevi, P. (2007). Nickel (II) determination by spectrophotometry coupled with preconcentration technique in water and alloy samples. Bulletin-Korean Chemical Society, 28(3), 373. https://doi.org/10.5012/bkcs.2007.28.3.373 DOI: https://doi.org/10.5012/bkcs.2007.28.3.373
Shah, M., Hameed, A., Kashif, M., Majeed, N., Muhammad, J., Shah, N., Rehan, T., Khan, A., Uddin, J., Khan, A., & Khan, H. (2024). Advances in agar-based composites: A comprehensive review. Carbohydrate Polymers, 346, 122619. https://doi.org/10.1016/j.carbpol.2024.122619 DOI: https://doi.org/10.1016/j.carbpol.2024.122619
Sharif, K., & Amital, H. (2024). Heavy metals in autoimmune diseases: too much noise in autoimmunity. Autoimmune Disorders, Chapter 19, 201-223. https://doi.org/10.1002/9781119858430.ch19 DOI: https://doi.org/10.1002/9781119858430.ch19
Shi, T., Jia, S., Chen, Y., Wen, Y., Du, C., Guo, H., & Wang, Z. (2009). Adsorption of Pb (II), Cr (III), Cu (II), Cd (II) and Ni (II) onto a vanadium mine tailing from aqueous solution. Journal of Hazardous Materials, 169(1-3), 838-846. https://doi.org/10.1016/j.jhazmat.2009.04.020 DOI: https://doi.org/10.1016/j.jhazmat.2009.04.020
Topuz, B., Adanur, Ş. M., & Yalcuk, A. (2017). A new method for simultaneous determination of trace amounts of Cu (II) and Ni (II) ions by preconcentration and spectrophotometric analysis. Turkish Journal of Chemistry, 41(5), 619-629. https://doi.org/10.3906/kim-1610-25 DOI: https://doi.org/10.3906/kim-1610-25
Trus, I., Gomelya, M., & Kryzhanovska, Y. (2023). The use of coagulants from industrial waste in water treatment processes. Journal of Chemical Technology and Metallurgy, 58(1), 178-186.
Ungureanu, E. L., & Mustatea, G. (2022). Toxicity of heavy metals. In: Environmental impact and remediation of heavy metals: IntechOpen. https://doi.org/10.5772/intechopen.102441 DOI: https://doi.org/10.5772/intechopen.102441
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Ajmal Shah, Balqees Khan, Adnan Khalid, Nimra Ali, Saroosh Wazir, Ume Aiman, Syed Adnan, Amna Yousaf, Maaz Ahmad

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with this journal agree to the following terms:
1) Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
2) Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.
3) Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work.


