Biological activity of a benzene sulfonamide on perfusion pressure and coronary resistance using an isolated rat heart model
DOI:
https://doi.org/10.14295/bjs.v3i4.540Keywords:
benzenesulfonamide, derivatives, perfusion pressure, dockingAbstract
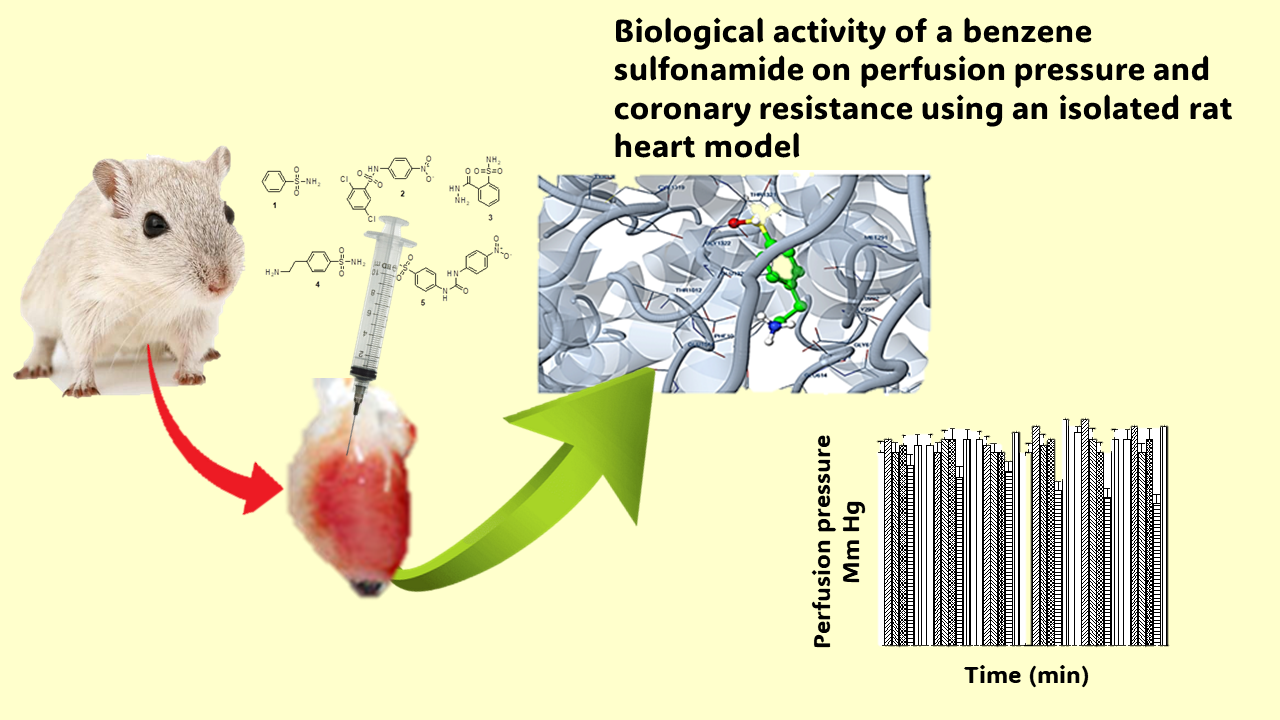
There are studies which indicate that some sulfonamide derivatives can produce changes in the cardiovascular system; however, their biological activity on perfusion presure and coronary resistance is not clear. The aim of this research was to evaluate the effect exerted by benzenesulfonamide, and their derivatives (2,5-dichloro- N-(4-nitro-phenyl)-benzene-sulfonamide, 2-hydrazino-carbonyl-benzenesulfonamide, 4-(2-amino-ethyl)-benze- ne-sulfonamide, and 4-[3-(4-nitro-phenyl)-ureido]-benzene- sulfonamide) on perfusion pressure and coronary reistance. To evaluate the biological activity of benzenesulfonamide and their derivatives on perfusion pressure and coronary reistance an isolated rat heart model was used. Furthermore, theoretical interaction of 4-(2-amino-ethyl)-benzenesul- fonamide with Calcium channel surface was determined using 6jp5 protein, nifedipine, amlodipine, verapamil and BayK 8644 as theoretical tools in a DockingServer program. The Results showed that 4-(2-amino-ethyl)-ben- zenesulfonamide decreased perfusion pressure and coronary resistance compared to benzenesulfonamide, 2,5-dichloro- N-(4-nitro-phenyl)-benzene-sulfonamide, 2-hydrazinocar- bonyl-benze-nesulfonamide, 4-[3-(4-nitro-phenyl)-ureido]- benenesulfonamide and the control conditions. Besides, theoretical data suggest that 4-(2-aminoethyl)benzenesulfo- namide could interact with aminoacid residues such as Glu614 and Ala320 involved in 6jp5 protein surface. This phenomenon could result in an ligand-Calcium channel complex formation to produce a decrease in perfusion pressure and vascular resistance. It is noteworthy that biological and experimental models used in this study is an invaluable research tool for investigating questions across the spectrum of physiologic functions of cardiovascular system such as perfusion pressure and coronary resistance
References
Archana, G., Arunkumar, R., & Omkumar, R. (2022). Assays for L-type voltage gated calcium channels. Analytical Biochemistry, 656, 114827. https://doi.org/10.1016/j.ab.2022.114827 DOI: https://doi.org/10.1016/j.ab.2022.114827
Azis, M., Ejaz, S., Tamam, N., Siddique, F., Riaz, N., Qais, F., & Iqbal, J. (2022). Identification of potent inhibitors of NEK7 protein using a comprehensive computational approach. Scientific Reports, 12, 1-17. https://doi.org/10.1038/ s41598-022- 10253-5 DOI: https://doi.org/10.1038/s41598-022-10253-5
Bergson, P., Lipkind, G., Lee, S., Duban, M., & Hanck, D. (2022).Verapamil block of T-type calcium channels. Molecular Pharmacology, 79(3), 411-419. https://doi.org/10.1124/mol.110.069492 DOI: https://doi.org/10.1124/mol.110.069492
Bisht, N., Sah, A., Bisht, S., & Joshi, H. (2021). Emerging need of today: significant utilization of various databases and softwares in drug design and development. Mini Reviews in Medicinal Chemistry, 21(8), 1025-1032. https://doi.org/10.2174/1389557520666201214101329. DOI: https://doi.org/10.2174/1389557520666201214101329
Cauich-Carrillo, R., Figueroa-Valverde, L., Rosas-Nexticapa, M., Garcimarrero-Espino, A., Lopez-Ramos, M., Mateu-Armad, V., Alvarez-Ramirez, M., & Lopez-Gutierrez, T. (2021). Evaluation of effect produced by a steroid-azetidine derivative against infarct area using a heart failure model. Parana Journal of Science and Education, 7(5), 31-39. https://sites.google.com/site/pjsciencea.
Chalkha, M., Akhazzane, M., Moussaid, F., Daoui, O., Nakkabi, A., Bakhouch. A., & El-Yazidi, M. (2022). Design, synthesis, characterization, in vitro screening, molecular docking, 3D-QSAR, and ADME-Tox investigations of novel pyrazole derivatives as antimicrobial agents. New Journal of Chemistry, 46(6), 2747-2760. https://doi.org/10.1039/D1NJ05621B DOI: https://doi.org/10.1039/D1NJ05621B
Figueroa-Valverde, L., Diaz-Cedillo, F., & García-Cervera, E. (2013). Positive inotropic activity induced by a dehydroisoandrosterone derivative in isolated rat heart model. Archives of Pharmacal Research, 36, 1270-1278. DOI: https://doi.org/10.1007/s12272-013-0166-7
Figueroa-Valverde, L., Diaz-Cedillo, F., Diaz-Ku, E., & Camacho-Luis, A (2009) Effect induced by hemisuccinate of pregnenolone on perfusion pressure and vascular resistance in isolated rat heart. African Journal of Pharmacy and Pharmacology, 3, 234-241.
Figueroa-Valverde, L., Diaz-Cedillo, F., Lopez-Ramos, M., & Garcia-Cervera, E. (2011). Changes induced by estradiol-ethylenediamine derivative on perfusion pressure and coronary resistance in isolated rat heart: L-type calcium channel. Biomedical Papers, 155(1), 27-32. https://doi.org/10.5507/bp.2011.018 DOI: https://doi.org/10.5507/bp.2011.018
Figueroa-Valverde, L., Lopez, M., Alvarez, M., Rosas, M., Diaz, F., Mateu, v. (2023). Evaluation of coumarin and their derivatives as Janus Kinase-3 inhibitors using a theoretical model. Brazilian Journal of Science, 2(12), 106-117, https://doi.org/10.14295/bjs.v2i12.423 DOI: https://doi.org/10.14295/bjs.v2i12.423
Figueroa-Valverde, L., Rosas, M., Lopez, M., Diaz, F., Alvarez, M., Mateu, V., & Melgarejo, M. (2023). Effect Produced by a Cyclooctyne Derivative on Both Infarct Area and Left Ventricular Pressure via Calcium Channel Activation. Drug Research, 73(2), 105-112. https://doi.org/DOI: 10.1055/a-1967-2004 DOI: https://doi.org/10.1055/a-1967-2004
Figueroa-Valverde, L., Rosas-Nexticapa, M., Alvarez-Ramirez, M., López-Ramos, M., Díaz-Cedillo, F., & Mateu-Armad, V. (2023). Evaluation of Biological Activity Exerted by Dibenzo [b, e] Thiophene-11 (6H)-One on Left Ventricular Pressure Using an Isolated Rat Heart Model. Drug Research, 73(5), 263-270. https://doi.org/10.1055/a-1995-6351 DOI: https://doi.org/10.1055/a-1995-6351
Figueroa-Valverde, L., Rosas-Nexticapa, M., Melgarejo, M., Díaz-Cedillo, D., López-Ramos, M., & Alvarez-Ramirez, M. (2023). Synthesis and Theoretical Interaction of 3-(2-oxabicyclo [7.4. 0] trideca-1 (13), 9, 11-trien-7-yn-12-yloxy)-steroid Deriva-tive with 17β-hydroxysteroid Dehydrogenase Enzyme Surface”, Biointerface Research in Applied Chemistry, 13(3), 1-11. https://doi.org/10.33263/BRIAC133.266 DOI: https://doi.org/10.33263/BRIAC133.266
Hess, D. A., Sisson, M. E., Suria, H., Wijsman, J., Puvanesasingham, R., Madrenas, J., & Rieder, M. J. (1999). Cytotoxicity of sulfonamide reactive metabolites: apoptosis and selective toxicity of CD8+ cells by the hydroxylamine of sulfamethoxazole. The FASEB Journal, 13(13), 1688-1698. https://doi.org/10.1096/fasebj.13.13.1688 DOI: https://doi.org/10.1096/fasebj.13.13.1688
Gökce, H., Öztürk, N., Sert, Y., El‐Azab, A., AlSaif, N., & Abdel‐Aziz, A. (2018). 4‐[(1, 3‐Dioxoisoindolin‐2‐yl)
methyl] benzenesulfonamide: Full Structural and Spectroscopic Characterization and Molecular Docking with Carbonic Anhydrase II. ChemistrySelect, 3(35), 10113-10124. https://doi.org/10.1002/slct.201802484 DOI: https://doi.org/10.1002/slct.201802484
Grześk, E., Szadujkis Szadurska, K., Bloch Bogusławska, E., Wiciński, M., Malinowski, B., KołTan, S., & GrześK, G. (2017). 2, 4, 6 Trimethyl N [3 (trifluoromethyl) phenyl] benzenesulfonamide increases calcium influx in lipopolisaccharide-pre-treated arteries. Experimental and Therapeutic Medicine, 13(2), 766-770. DOI: https://doi.org/10.3892/etm.2016.3986
Hangeland, J., Cheney, D., Friends, T., Swartz, S., Levesque, P., Rich, A., & Ewing. W. (2008). Design and SAR of selective T-type calcium channel antagonists containing a biaryl sulfonamide core. Bioorganic & Medicinal Chemistry Letters, 18(2), 474-478. https://doi.org/10.1016/j.bmcl.2007.11.103 DOI: https://doi.org/10.1016/j.bmcl.2007.11.103
Harris, P., Boloor, A., Cheung, M., Kumar, R., Crosby, R., & Davis-Ward, R. Stafford, (2008). Discovery of 5-[[4-[(2, 3-dimethyl-2 H-indazol-6-yl) methylamino]-2-pyrimidinyl] amino]-2-methyl- benzenesulfonamide (Pazopanib), a novel and potent vascular endothelial growth factor receptor inhibitor. Journal of Medicinal Chemistry, 51(15), 4632-4640. https://doi.org/10.1021/jm800566m DOI: https://doi.org/10.1021/jm800566m
Kanda, Y., Kawanishi, Y., Oda, K., Sakata, T., Mihara, S., Asakura, K., & Konoike, T. (2001). Synthesis and structure-activity relationships of potent and orally active sulfonamide ETB selective antagonists. Bioorganic & Medicinal Chemistry, 9(4), 897-907. https://doi.org/10.1016/S0968-089600) 00305-9 DOI: https://doi.org/10.1016/S0968-0896(00)00305-9
Kim, J., Keum, G., Chung, H., & Nam, G. (2016). Synthesis and T-type calcium channel-blocking effects of aryl (1, 5-disubstituted-pyrazol-3-yl) methyl sulfonamides for neuropathic pain treatment. European Journal of Medicinal Chemistry, 123, 665-672. DOI: https://doi.org/10.1016/j.ejmech.2016.07.032
Krishnamurthy, V., Bohall, B., Kim, C., Moustakas, D., Christianson, D., & Whitesides, V. (2007). Thermodynamic parameters for the association of fluorinated benzenesulfonamides with bovine carbonic anhydrase II. Chemistry-An Asian Journal, 2(1), 94-105. https://doi.org/10.1002/asia.200600360 DOI: https://doi.org/10.1002/asia.200600360
Kumar, S., Rajput, M., Tickoo, S. (2021). Laws, Regulations, Policies and Guidelines Governing the Care and Use of Laboratory Animals. Springer, Singapore, 23-38, 2021. DOI: https://doi.org/10.1007/978-981-16-0987-9_2
Leblond, C., & Hoff H. (1994). Effect of sulfonamides and thiourea derivatives on heart rate and organ morphology. Endocrinology, 35(4), 229-233. https://doi.org/10.1210/endo-35-4-229 DOI: https://doi.org/10.1210/endo-35-4-229
Lehr, D. (1957). Clinical toxicity of sulfonamides. Annals of the New York Academy of Sciences, 69(3), 417-447. DOI: https://doi.org/10.1111/j.1749-6632.1957.tb49682.x
Mahanthesh, M., Ranjith, D., Yaligar, R., Jyothi, R., Narappa, G., &. Ravi, M. (2020). Swiss ADME prediction of phytochemicals present in Butea monosperma (Lam.) Taub. Journal of Pharmacognosy and Phytocheemistry, 9(3), pp. 1799-1809.
Malysz, J., Grønlien, J., Anderson, J., Håkerud, M., Thorin-Hagene K., Ween, K.,, & Gopalakrishnan M. (2009). In vitro pharmacological characterization of a novel allosteric modulator of α7 neuronal acetylcholine receptor, 4-(5-(4-chlo- rophenyl)-2-methyl-3-propionyl-1H-pyrrol-1-yl) benzenesulfona- mide (A-867744), exhibiting unique pharmacological profile. Journal of Pharmacology and Experimental Therapeutics, 330(1), 257-267, https://doi.org/10.1124/jpet.109.151886 DOI: https://doi.org/10.1124/jpet.109.151886
Manson, R., Marche, P., & Hintze, T. (2003). Novel vascular biology of third-generation L-type calcium channel antagonists: ancillary actions of amlodipine. Arteriosclerosis, Thrombosis, and Vascular Biology, 23(12), 2155-2163. https://doi.org/10.1161/01.ATV.0000097770.66965.2A DOI: https://doi.org/10.1161/01.ATV.0000097770.66965.2A
Nelson, K., Christopher, F., &Milton, N. (2022). Teach yourself SPSS and STATA. International Journal of Academic Pedagogical Research, 7(6), 84-122.
Nguyen, P., Elkamhawy, A., Choi, Y., Lee, C., Lee, K., & Cho, J. (2022). Suppression of Tumor Growth and Cell Migration by Indole-Based Benzenesulfonamides and Their Synergistic Effects in Combination with Doxorubicin. International Journal of Molecular Sciences, 23(17), 1-23. https://doi.org/10.3390/ijms23179903 DOI: https://doi.org/10.3390/ijms23179903
Nie, Y., Yang, J., Zhou, L., Yang, Z., Liang, J., Liu, Y., & Zhang, Y. (2022). Marine fungal metabolite butyrolactone I prevents cognitive deficits by relieving inflammation and intestinal microbiota imbalance on aluminum trichloride-injured zebrafish. Journal of Neuroinflammation, 19(1), 1-17. https://doi.org/10.1186/s12974-022-02403-3 DOI: https://doi.org/10.1186/s12974-022-02403-3
Olda, A., Trixie, J., Bolang, G., Witular, Y., & Langi, S. (2022). Nifedipine, Calcium Channel Blocker (Antihypertensive), as a Tocolytic to inhibit Premature Birth in Reducing the Risk of Neonatal Death in Childbirth: Meta-Analysis and Systematic Review of Large Clinical Trial. Indonesian Journal of Obstetrics and Gynecology, 1, 58-62. https://doi.org/10.32771/inajog.v10i1.1549 DOI: https://doi.org/10.32771/inajog.v10i1.1549
Pervaiz, M., Riaz, A., Munir, A., Saeed, Z., Hussain, S., Rashid, A., Umar, Y., & Adnan, A. (2010) Synthesis and characterization of sulfonamide metal complexes as antimicrobial agents. Journal of Molecular Structure, 1202, 1-35. https://doi.org/10.1016/j.molstruc.2019. 127284 DOI: https://doi.org/10.1016/j.molstruc.2019.127284
Phopin, K., Ruankham, W., Prachayasittikul, S., Prachayasittikul, V., & Tantimongcolwat, T. (2020). Insight into the molecular interaction of cloxyquin (5-chloro-8-hydroxyquinoline) with bovine serum albumin: biophysical analysis and computational simulation. International Journal of Molecular Sciences, 21, 1-18. https://doi.org/10.3390/ijms21010249 DOI: https://doi.org/10.3390/ijms21010249
Ren, Y., Ma, Y., Cherukupalli, S., Tavi, J., Menéndez-Arias, L., Liu, X., Zhan, p. (2020). Discovery and optimization of benzenesulfonamides-based hepatitis B virus capsid modulators via contemporary medicinal chemistry strategies. European Journal of Medicinal Chemistry, 206, 1-13. https://doi.org/10.1016/j.ejmech.2020.112714 DOI: https://doi.org/10.1016/j.ejmech.2020.112714
Roy, A., Sarkar, T., Datta, S., Maiti, A., Chakrabarti, M., Mondal, T., & Roy, K. (2022). Structure‐based discovery of (S)‐2‐amino‐6‐(4‐fluorobenzyl)‐5,6,11,11a‐tetrahydro‐1H‐imida- zo[1′,5′:1,6]pyrido[3,4‐b]indole‐1, 3 (2H)‐dione as low nanomolar, orally bioavailable autotaxin inhibitor. Chemical Biology & Drug Design, 99(3), 496-503. https://doi.org/10.1111/cbdd.14017 DOI: https://doi.org/10.1111/cbdd.14017
Schwartz, W., Relman, A., & Leaf, A. (1995). Oral administration of a potent carbonic anhydrase inhibitor (" Diamox"). III. Its use as a diuretic in patients with severe congestive heart failure due to cor pulmonale. Annals of Internal Medicine, 42(1), 79-89. https://doi.org/10.7326/0003-4819-42-1-79 DOI: https://doi.org/10.7326/0003-4819-42-1-79
Shao, P., Ye, F., Chakravarty, P., Varughese, D., Herrington, J., Dai, G., & Duffy, J. (2012). Aminopiperidine sulfonamide Cav2. 2 channel inhibitors for the treatment of chronic pain. Journal of Medicinal Chemistry, 55(22), 9847-9855. https://doi.org/10.1021/jm301056k DOI: https://doi.org/10.1021/jm301056k
Takenaka, T., Shiono, K., Honda, K., Asano, M., Miyazaki, I., & Maeno, H. (1982). Antihypertensive and adrenoceptor blocking properties of new sulfonamide-substituted phenylethylamines. Clinical and Experimental Hypertension, 4(1-2), 125-137. https://doi.org/10.3109/10641968209061580 DOI: https://doi.org/10.3109/10641968209061580
Talley, J., Brown, D., Carter, J., Graneto, M., Koboldt, C., Masferrer, J., & Seibert, K. (2000). 4-[5-Methyl-3-phenylisoxazol-4-yl]-benzene-sulfonamide, valdecoxib: a potent and selective inhibitor of COX-2. Journal of Medicinal Chemistry, 43(5), 775-777. https://doi.org/10.1021/jm990577v DOI: https://doi.org/10.1021/jm990577v
Tellew, J., Baska, R., Beyer, S., Carlson, K., Cornelius, L., Fadnis, L., & Macor, J. (2003). Discovery of 4′-[(Imidazol-1-yl) methyl] biphenyl-2-sulfonamides as dual endothelin/Angiotensin II receptor antagonists. Bioorganic & Medicinal Chemistry Letters, 13(6), 1093-1096. https://doi.org/10.1016/S0960-894X(03)00018-0 DOI: https://doi.org/10.1016/S0960-894X(03)00018-0
Tilton. R., Munsch, C., Sherwood, S., Chen, S., Chen, Y., Wu, C., & Brock, T. (2000). Attenuation of pulmonary vascular hypertension and cardiac hypertrophy with sitaxsentan sodium, an orally active ETAreceptor antagonist. Pulmonary Pharmacology & Therapeutics, 13(2), 87-97. https://doi.org/10.1006/pupt.2000. 0237 DOI: https://doi.org/10.1006/pupt.2000.0237
Turner, C., Pawluk, M., Bolsoni, J., Zeglinski, M., Shen, Y., Zhao, H., & Granville, D. J. (2022). Sulfaphenazole reduces thermal and pressure injury severity through rapid restoration of tissue perfusion. Scientific Reports, 12(1), 12622. https://doi.org/10.1038/s41598-022-16512-9 DOI: https://doi.org/10.1038/s41598-022-16512-9
Vilter, C., & Blankenhorn, M. (1944). The toxic reactions of the newer sulfonamides. Journal of the American Medical Association, 126(11), 691-695. doi:10.1001/jama.1944.02850460021005 DOI: https://doi.org/10.1001/jama.1944.02850460021005
Wu, C., Chan, M., Stavros, F., Raju, B., Okun, I., Mong, S., & Dixon, R. (1997). Discovery of TBC11251, a potent, long acting, orally active endothelin receptor-A selective antagonist. Journal of Medicinal Chemistry, 40(11), 1690-1697. https://doi.org/10.1021/jm9700068 DOI: https://doi.org/10.1021/jm9700068
Zhang, Q., Xia, Z. Joshi, S., Scott, V., &. Jarvis, M. (2015). Optimization of ADME properties for sulfonamides leading to the discovery of a T-type calcium channel blocker, ABT-639. Medicinal Chemistry Letters, 6(6), 641-644. https://doi.org/10.1021/acsmedchemlett.5b00023 DOI: https://doi.org/10.1021/acsmedchemlett.5b00023
Zubrienė, A., Smirnov, A., Dudutienė, V., Timm, D., Matulienė, J., Michailovienė, V., & Matulis, D. (2017). Intrinsic Thermodynamics and Structures of 2, 4‐and 3, 4‐Substituted Fluorinated Benzenesulfonamides Binding to Carbonic Anhydrases. ChemMedChem. 12(2), 161-176. https://doi.org/10.1002/cmdc.201600509 DOI: https://doi.org/10.1002/cmdc.201600509
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Magdalena Alvarez-Ramirez, Lauro Figueroa-Valverde, Marcela Rosas-Nexticapa, Maria López-Ramos, Maria Virginia Mateu-Armad, Eli Alejandra Garcimarrero-Espino, Lenin Hau-Heredia, Regina Cauich-Carrillo, Julliete Mijangos-Sanchez

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with this journal agree to the following terms:
1) Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
2) Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.
3) Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work.


